Imagine a cluster of living brain cells in a dish that can learn to play a simple video game. It sounds like science fiction, but scientists at the University of California, Santa Cruz (UCSC) have recently achieved exactly that. They trained lab-grown brain organoids – tiny 3D tissues made of neurons – to control a cart-pole balancing game (a bit like balancing a digital broomstick) using nothing but electrical signals as feedback. The lab-grown mini-brains improved dramatically at the task, boosting their success rate from about 4.5% to 46% after targeted training.
This breakthrough marks the first time researchers have demonstrated goal-directed learning in brain organoids, opening the door to “biological computers” that learn and adapt like a real brain. In this article, we’ll explain what it means for brain cells to play a video game, how the UCSC experiment worked, why the cart-pole test is important, and how this advance could impact neuroscience, computing, and more.
What does it mean for lab-grown brain cells to “play a video game”?
When lab-grown brain cells “play a video game,” they are not consciously using a controller. Instead, a brain organoid—a small sphere of neural tissue—is interfaced with a computer simulation. The organoid receives data about the game state via electrical signals from electrodes and responds with neural firing patterns that the computer interprets as game actions. In this setup, the neurons act as the controller.
In a UCSC experiment, organoids “played” a virtual cart-pole balancing task. The organoid adjusted its firing to correct the pole’s balance. Learning is achieved through a closed-loop system: if the organoid performs well, it continues undisturbed; if it fails, it receives an electronic “nudge” or feedback signal. This demonstrates that even simplified 3D neural structures can process real-time information and adapt to goal-oriented tasks, building on previous 2D neural culture experiments like those involving the game Pong.
UCSC brain organoids cart-pole experiment explained
In February 2026, a UCSC team led by Ash Robbins, Mircea Teodorescu, and David Haussler announced they had trained a peppercorn-sized brain organoid, grown from mouse stem cells, to solve the cart-pole problem. The organoid, containing millions of neurons, sat on a microelectrode chip that functioned as a bioelectrical interface.
The computer encoded the angle and direction of a falling virtual pole into electrical impulses sent to the organoid. The organoid’s neural responses were then translated into instructions to move the cart left or right. The researchers used a closed-loop coaching regime: an AI-based reinforcement learning algorithm monitored performance. If the organoid’s balance time didn’t improve every five episodes, the algorithm delivered a targeted electrical “punishment” signal to specific neurons to encourage rewiring. This method increased the organoid’s success rate from a baseline of 4.5% to 46%, proving the tissue could be coached to perform a complex control task.

Cart-pole balancing test: why AI researchers use it
The cart-pole balancing test, or inverted pendulum problem, is a classic benchmark in robotics and AI. It requires a controller to keep a hinged pole upright by moving a cart. It is a staple of reinforcement learning (such as OpenAI Gym’s “CartPole-v0”) because it tests continuous control and real-time feedback processing.
While simple to set up, it is difficult to solve, requiring constant small corrections. Humans solve similar problems when learning to stand or walk. Using this benchmark allowed UCSC researchers to compare the organoid’s biological learning against random chance and artificial intelligence. The organoid’s improvement showed it could adapt its output in a goal-directed way, which is a fundamental step toward more complex tasks.
How brain organoids were trained using electrical reward and punishment signals
Training organoids involves translating reward and punishment into electrical signals. In the UCSC study, “reward” was the absence of disruptive input—the organoid was left alone when performing well. “Punishment” consisted of a targeted electrical stimulus delivered at the end of a failed episode.
The timing and targeting of these signals were crucial. An external reinforcement learning algorithm acted as a “coach,” identifying which neural circuits likely caused the failure and perturbing them with a jolt. This encouraged the network to explore different activity patterns. Because this process relies on electrical activity to drive synaptic changes (plasticity), the organoid learned to balance the pole without the need for biological reward chemicals like dopamine.

Brain organoids success rate jump from 4.5% to 46% — what changed?
The jump in success from 4.5% to 46% was driven by the shift from random or non-specific stimulation to adaptive, targeted coaching. Without meaningful guidance, organoids only succeeded by luck at a near-chance rate. With the reinforcement learning algorithm providing specific feedback based on neural activity, the signals carried information the organoid could use to improve.
The consistency of the rules—no disturbance for success and a targeted jolt for failure—allowed the organoid to maximize the “no disturbance” state. While the 46% success rate is lower than a well-trained AI or human, it represents a tenfold improvement over the untrained state. This demonstrates that while the organoid has inherent plasticity, it requires a structured “language” of feedback to unlock its learning potential.
How lab-grown neurons learn without eyes, ears, or sensory input
Brain organoids lack sensory organs and modulatory chemicals like dopamine, yet they learn by using electrodes as substitute “senses.” The computer translates game data (like the pole’s tilt) directly into electrochemical pulses, which the neurons process as if they were receiving natural sensory input.
Learning occurs because the capacity to adapt is intrinsic to neural tissue. The organoid responds to the external patterns of stimulation provided by the researchers, forming new circuits to avoid disruptive feedback. This “hacked” learning process demonstrates that sensory-motor loops can be artificially engineered. Despite having no awareness, the organoid functions as a biological participant in a simulated environment, showing that neural tissue can adapt to its environment as long as it is interfaced with a reliable input-output structure. still self-organize to meet a goal.
Brain organoids vs AI reinforcement learning: key differences
While brain organoids and AI both use feedback loops, they differ significantly in their mechanics. AI learns through numerical weight adjustments and algorithms like backpropagation, whereas organoid learning is an emergent biological process involving synaptic reorganization. AI systems can be trained at superhuman speeds and cloned, but organoids operate in real-time and require rest. However, biology is vastly more energy-efficient; the human brain uses approximately 20 watts, while silicon supercomputers require millions of watts for comparable tasks.
In terms of memory, AI retains knowledge indefinitely, while the UCSC organoid showed poor long-term retention, returning to baseline performance after a 45-minute rest. AI networks can scale to billions of optimized parameters, whereas organoids are limited to around a million neurons, though each biological neuron is individually more complex than an artificial node. AI is reproducible and deterministic, but biological systems are unique, variable, and difficult to inspect. Finally, AI is explicitly programmed to maximize a reward function, whereas an organoid’s motivation is externally imposed via stimulation patterns; the organoid reacts to stimuli rather than internally computing a strategy to “win.”
What are brain organoids made from and how are they grown?
Brain organoids, or “mini-brains,” are simplified 3D versions of brain tissue grown from stem cells, such as induced pluripotent stem cells (iPSCs) or embryonic stem cells. Scientists use biochemical cues and 3D cultures—often involving spinning bioreactors or gels—to encourage these cells to self-organize into structures that resemble the developing brain. Over weeks or months, they form layered structures and fluid-filled cavities called ventricles.
The UCSC study used organoids grown from mouse stem cells, though human cells have been used since 2013 (pioneered by Madeline Lancaster) to model diseases like Alzheimer’s. These organoids are limited to a few millimeters in size because they lack blood vessels, but they can contain several million neurons, comparable to a small insect brain. While they produce neural firing and brain wave patterns, they are not conscious. They serve as research tools that can be interfaced with chips to act as computational components.

Bioelectrical interfaces: how computers connect to living brain tissue
The connection between computers and organoids is facilitated by microelectrode arrays (MEAs) or bioelectronic chips, such as the high-density chip from Maxwell Biosystems used by UCSC. This interface allows for a two-way “closed-loop” highway: electrodes record voltage changes when neurons fire and deliver small electrical currents to stimulate them.
In the UCSC experiment, the computer translated the game state into spatialized electrical patterns—stimulating certain areas based on the pole’s tilt—and decoded the organoid’s firing patterns as game actions. Advances in 3D bioelectronic arrays, such as those at Northwestern University, aim to record from hundreds of points to map neural conversations in real-time. These interfaces are essential because they turn a static clump of cells into an interactive biological processor, similar to technology used in brain-machine interfaces for prosthetics.
Could living brain cells power future computers or hybrid AI systems?
“Biological computing” explores using living cells, which are massively parallel and self-organizing, to perform tasks. Because the human brain is so energy-efficient compared to supercomputers like Frontier, researchers like Dr. Thomas Hartung suggest “biocomputers” could eventually surpass silicon in learning and handling uncertainty. Startups like Cortical Labs have already developed the “DishBrain” and “CL1” systems, which allow programmers to treat living neurons as a computing resource via a cloud API to play games like Pong and Doom.
Future hybrid systems might combine the rapid arithmetic of silicon with the adaptive, analog processing of biology. Organoids could be used for “reservoir computing” or speech recognition. While biological computing faces challenges regarding reliability, longevity, and scalability, it offers potential for robotic control and more efficient drug testing. Though not intended to replace standard CPUs immediately, these developments suggest that hybrid bio-digital computers may eventually outperform either system alone.

“Biological computing” and “organoid intelligence” definitions people are searching
Biological computing refers to using biological molecules or systems—such as neurons, DNA, or proteins—to perform computational tasks. Instead of silicon bits, information is processed via biochemistry and neural firing. This field aims to combine the parallelism and learning efficiency of biology with technological programmability.
Organoid intelligence (OI) is a specific subset of biological computing that focuses on using 3D cultured brain organoids coupled with machine interfaces as “wetware.” The goal is to create systems that can learn, remember, and perform AI-like functions. The term gained prominence following a 2023 roadmap by scientists aiming to develop brain-like computers. OI represents a biological form of AI—intelligence cultivated from living neurons rather than artificial code. The UCSC cart-pole experiment is a primary example of this emerging field.
Ethics of lab-grown brain cells in computing and gaming experiments
The use of living brain cells as computational tools raises several ethical concerns. A primary question is whether these organoids could achieve sentience or consciousness. While they currently resemble fetal tissue and lack necessary structures for complex thought, researchers suggest monitoring complexity to stay below a sentience threshold. The use of human-derived cells adds philosophical weight, questioning the rights of donors and the morality of using human neural tissue for “gaming.”
While the UCSC study used mouse cells, previous experiments have used human neurons. Ethicists are concerned about the welfare of the tissue, noting that even if pain receptors aren’t currently present, future complex models may require animal-like welfare protocols. There is also a fear of the “slippery slope,” where minor experiments could lead to “brains in vats” or human-animal chimeras used for menial tasks. David Haussler emphasized that the goal is medical research, not replacing silicon CPUs, as using human organoids as computing elements is ethically fraught. Current regulations are also in a “gray area,” as organoids fall between human subject research and animal testing.
Doom vs Pong vs cart-pole: the most famous “brain cells playing games” milestones
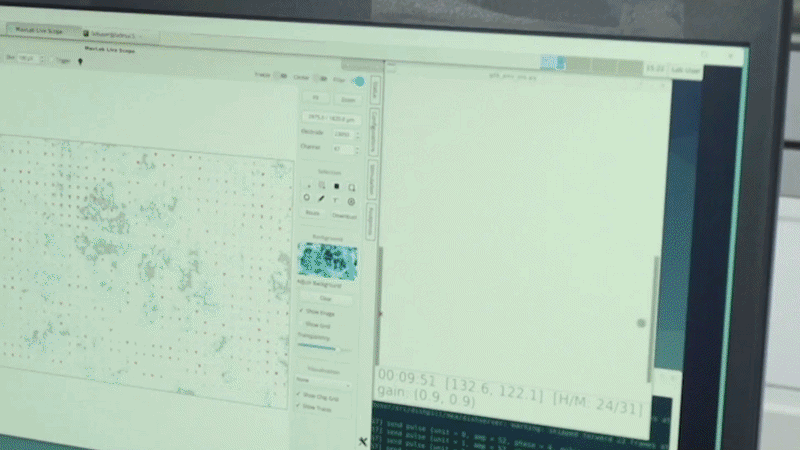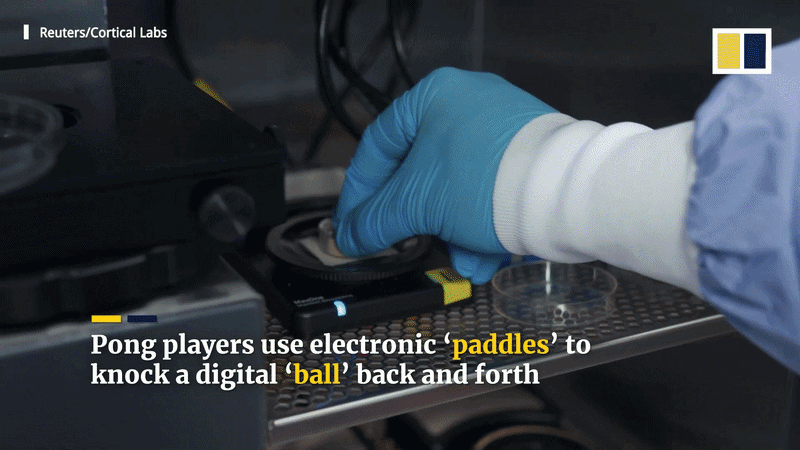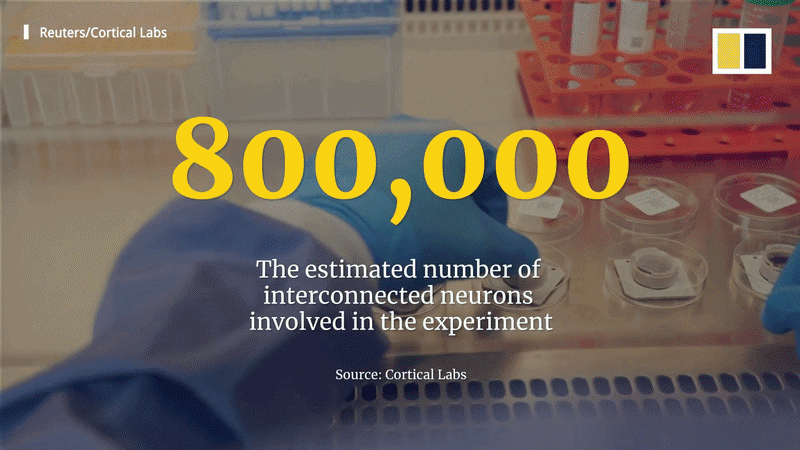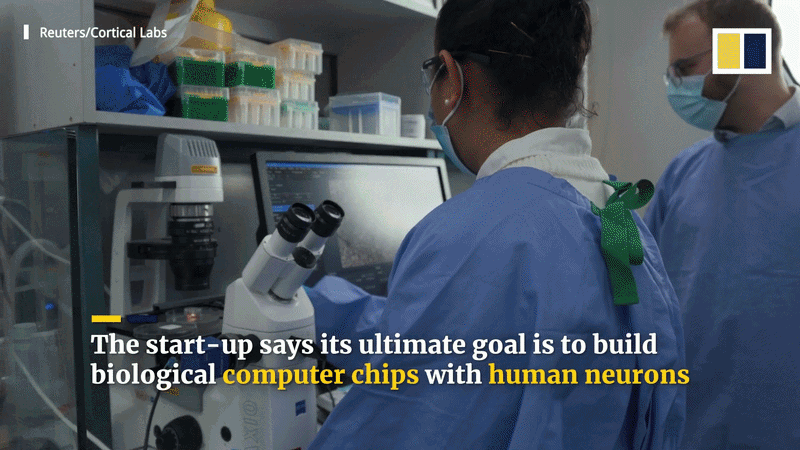
The field has evolved through several key milestones. In 2022, Cortical Labs’ “DishBrain” used 800,000 neurons (human and mouse) to play a 2D version of Pong, achieving a 70% success rate. By 2023-2024, the CL1 system performed basic tasks in the 3D first-person shooter DOOM, with human neurons learning to move and fire at enemies. While the performance was rudimentary, it showed the approach could scale to complex environments.
The 2026 UCSC cart-pole experiment is significant as the first peer-reviewed demonstration of learning in a 3D organoid. Unlike the earlier demos, it provided quantitative validation of a 10x success boost through rigorous RL coaching. These experiments build on precursors from the 2000s, such as Steve Potter’s work with neurons in flight simulators. Together, these milestones represent a progression from simple sensory response (Pong) to complex navigation (Doom) and controlled, adaptive learning (Cart-pole).
What this breakthrough could mean for neuroscience research and brain disease modeling
The ability to train organoids provides a controlled lab model for studying the biological mechanics of learning and neuroplasticity. Researchers can observe gene expression and synaptic changes in real-time. This has massive implications for disease modeling; scientists can grow organoids from the stem cells of patients with Alzheimer’s, autism, or ADHD to see how specific mutations affect learning ability.
These models also serve as a platform for testing therapeutics. Instead of starting with animal models, researchers can apply drugs to organoids and use their “game performance” (like balance time) as a quantifiable readout for cognitive improvement. The breakthrough also encourages the development of “assembloids”—interconnected organoids simulating different brain regions. Furthermore, it bridges AI and neuroscience, as biological learning might inspire new “neuromorphic” AI architectures. In the future, “brain-on-chip” technology could even allow for personalized medicine, using a patient’s own cells to predict how their brain might respond to specific treatments.

Biggest limitations right now: performance, scaling, and reliability
Despite recent successes, biological computing remains in its infancy. Performance is a major bottleneck; AI algorithms and human toddlers still vastly outperform organoids, which achieved only 46% success at a simple task. Stability is another issue, as UCSC organoids showed poor long-term retention, returning to baseline performance after short breaks. Scaling is physically limited because organoids lack blood vessels, causing internal cells to die if they grow too large.
Biological systems are also inconsistent; unlike cloned AI models, no two organoids are identical, making results difficult to reproduce. Training is complex and lacks a “compiler,” requiring tedious manual tuning of electrical signals. Furthermore, organoids have finite lifespans and require intensive environmental maintenance (nutrients, temperature control). Finally, current biointerfaces act as a bandwidth bottleneck, capturing only a fraction of the millions of neural conversations occurring within the tissue. Because of these factors, conventional computers are not in danger of being replaced by “brain chips” anytime soon.
Frequently Asked Questions (FAQs)
- What are lab-grown brain cells or brain organoids?
These are 3D clusters of neurons, often called “mini-brains,” grown from stem cells. They fire electrical signals and mimic early developmental brain stages at a size of 3-5 millimeters. - How can they “play” without eyes or a body?
Researchers use an electrode loop to convert game info into electrical pulses. The organoid’s responses are translated into game actions, serving as a living processor. - Are they conscious?
No. They lack sensory organs and pain receptors, reacting physically to stimuli via synaptic changes without internal awareness or a mind. - What tasks have been learned so far?
- Pong (2022): Neurons acted as a paddle to return a ball.
- Doom (2023/24): Human neurons performed basic character movement and firing.
- Cart-pole (2025/26): An organoid balanced a virtual pole with quantitative success.
- How are they trained?
Through electrical reinforcement learning. Corrective pulses are delivered for poor performance; improvement is rewarded by the absence of disturbance. - Why play games?
Games serve as benchmarks to test learning and information processing for research in neuroscience, AI, and biocomputing. - How do they compare to AI?
AI is faster and more stable. Organoids are proof-of-concept but demonstrate self-organized learning and extreme energy efficiency. - Could they power future computers?
“Organoid intelligence” (OI) envisions living neurons as biocomputers or hybrid processors to handle complex, fuzzy problems. - What are the ethical concerns?
Issues include potential future sentience, the use of human-derived cells, donor consent, and treating living tissue as a tool. - How far are we from a human-level lab brain?
Very far. Organoids have millions of neurons compared to the 86 billion in a human brain.

Conclusion
The UCSC cart-pole experiment demonstrates the problem-solving potential of biological neural networks. This achievement establishes adaptive biological computing, offering insights into brain disorders and memory. While current organoids are rudimentary, they prove that intelligence can emerge from biological substrates when provided with feedback loops. Future work will focus on scaling performance and the ethical navigation of tapping into nature’s wetware to learn and compute. a new chapter in the story of computing and understanding intelligence.
Sources and citation
- Cerf, E. (2026, February 19). Brain organoids can be trained to solve a goal-directed task. UCSC News.
- Marnell, B. (2026, March 2). Scientists Make Breakthrough That Allows Lab-Grown Brain Cells To Play A Sort Of Video Game. GameSpot.
- AZoLifeSciences. (2026, February 20). Lab-Grown Brain Tissue Masters a Classic Computing Benchmark. Reviewed by Lauren Hardaker.
- Landes, A. (2026, March 2). Scientists Teach Lab-Grown Brain Cells to Play Video Games. Gadget Review.
- Robbins, A., et al. (2026). Goal-directed learning in cortical organoids (publisher page). Cell Reports.
- Ledford, H. (2022, October 12). Neurons in a dish learn to play Pong — what’s next?. Nature News.
- UCL News. (2022, October 13). Human brain cells in a dish learn to play Pong.
- Tangermann, V. (2026, March 1). Researchers Get Human Brain Cells Running Doom. Futurism.
- Biological computer with real human neurons learns to shoot in Doom. Interesting Engineering.
- Organoid intelligence. Wikipedia. Accessed March 9, 2026.
- Biocomputing: the promise of biological computing. Polytechnique Insights. Published November 28, 2023.
- Smirnova, L., et al. (2023). Organoid intelligence (OI): the new frontier in biocomputing and intelligence-in-a-dish. Frontiers in Science.
- Cai, H., et al. (2023). Brain organoid reservoir computing for artificial intelligence. Nature Electronics.
- Kagan, B. J., et al. (2022). In vitro neurons learn and exhibit sentience when embodied in a simulated game-world (publisher page). Neuron (Cell Press).
Recommended
- Optimizing Blender Camera Settings with The View Keeper Add-on
- Substance Painter vs Mari: Which 3D Texturing Tool Is Best for You?
- VTuber Model Guide: Everything You Need to Know About Creating and Using VTuber Avatars
- Nobody Wants to Die Noir Retrofuturistic Detective Game: Trailer, Reviews & Player Feedback
- How Do I Add a Camera to My Blender Scene?
- Wind in Unreal Engine 5: WindDirectionalSource, Foliage Wind, Niagara Forces, Cloth, and Groom Hair Setup
- How do I set a camera to render in orthographic mode in Blender?
- How to Create Your Own Metahuman in Unreal Engine 5: Complete Step-by-Step Guide
- High-Level Metahuman Mocap Workflow: Professional Techniques for Unreal Engine 5 Animation
- Best Render Farm: Guide to Top Cloud Rendering Solutions for 3D Artists & Game Developers